Medical Imaging & Radiation Oncology
In Medical Imaging and Radiotherapy, researchers at MIBE work on novel technologies for computed tomography or magnetic resonance imaging, explore the radiobiological effects of novel radiotherapy concepts, and develop novel tracers and contrast agents with specific molecular sensitivity.
Phase-Contrast X-Ray Imaging
PI Franz Pfeiffer
Chair of Biomedical Physics
Publications
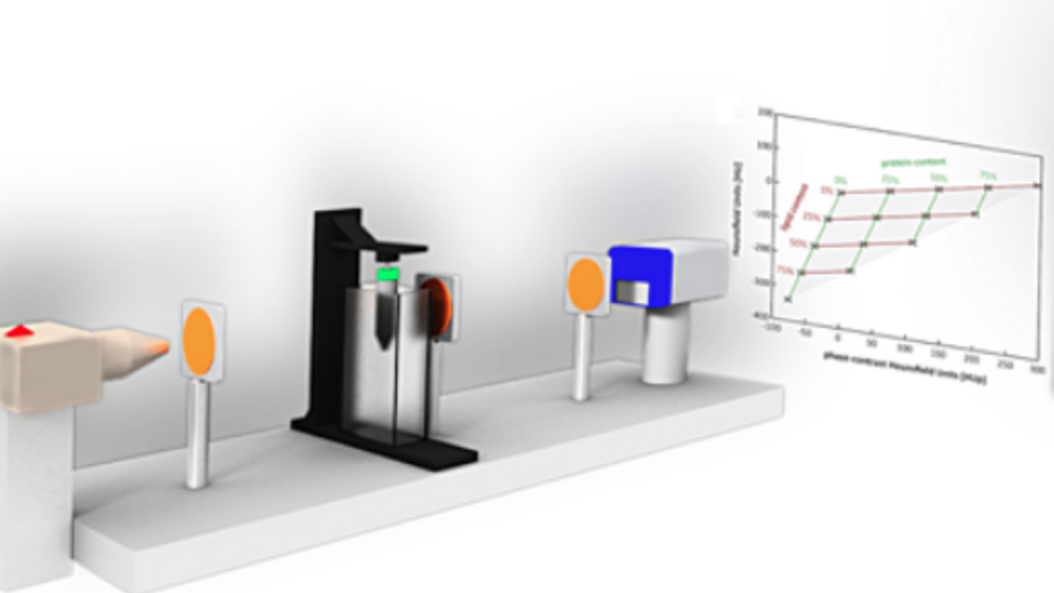
The basic physics principles of x-ray image formation in radiology have remained essentially unchanged since Röntgen first discovered x-rays over a hundred years ago. The conventional approach relies on x-ray attenuation as the sole source of contrast and ignores another, complementary source of contrast. Phase-contrast imaging techniques, on the other hand, offer ways to augment or complement standard attenuation contrast by incorporating phase information. In the recent past several developments have been made that now allow translating x-ray phase-contrast to future clinical applications in radiography and computed tomography.
Biomedical Imaging Physics
PI Julia Herzen
Research Group Biomedical Imaging Physics
Publications
The team around Prof. Herzen develops novel X-ray imaging methods using highly brilliant synchrotron radiation and conventional laboratory X-ray sources. They mainly focus on quantitative multi-modal approaches combining spectral and phase-contrast imaging. Currently, they are aiming at applying these methods for improved breast cancer detection and for quantitative 3D virtual histology of human tissue.
More information:
TEDx Talk (Youtube-Video): Novel X-Ray technology that can revolutionize preventive medicine
Nuclear Magnetic Resonance Imaging
PI Axel Haase
Munich Institute of Biomedical Engineering
Publications
Nuclear Magnetic Resonance (NMR) is a spectroscopic method, which is commonly used in science to elucidate the structure and dynamics of molecules. It uses strong magnetic fields (up to 23 Tesla) with highest homogeneity, temporal stability, and with radio frequency fields (up to 1 GHz). The additional use of magnetic field gradients (up to 1 Tesla/m) allows for the acquisition of MR images.
In general, Magnetic Resonance Imaging (MRI) uses the 1H MR signal from water and lipid molecules to create 2D or 3D images of biological objects (organs, animals, plants, human subjects). MRI is a non-invasive method and shows no biological side effects.
Therefore it is ideally suited to study and visualize the anatomy, function and metabolism of internal organs.
Theranostics
PI Wolfgang Weber
Professorship of Nuclear Medicine
Publications
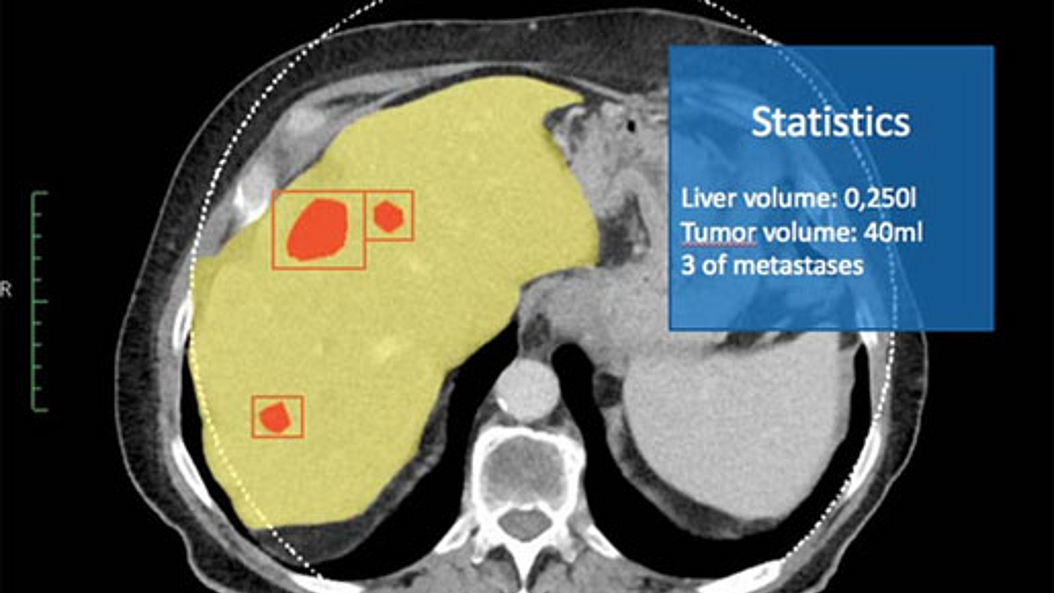
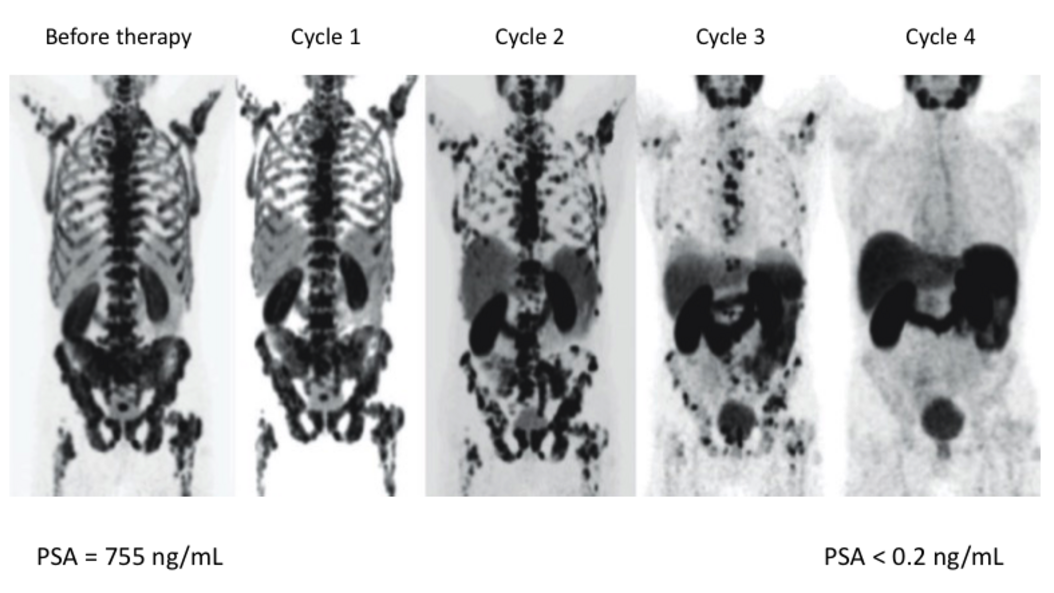
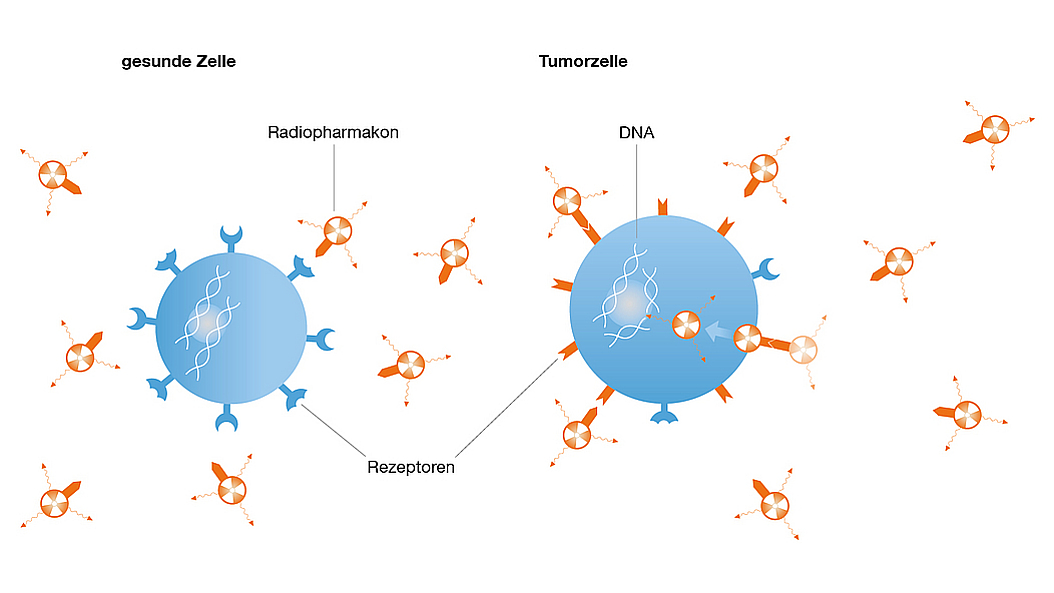
Theranostics, the combination of imaging and targeted radionuclide therapy, aims to treat cancer by radioisotopes that specifically accumulate in the tumor tissue due to molecular targeting. Theranostics expands the use of radiotherapy - one of the most effect therapies for virtually all cancers - to the treatment of patients with metastatic disease. Quantitative nuclear imaging is used to select patients for targeted radionuclide therapy, calculate radiation doses and monitor tumor response.
Biological & Optical Imaging
PI Vasilis Ntziachristos
Chair of Biological Imaging
Publications
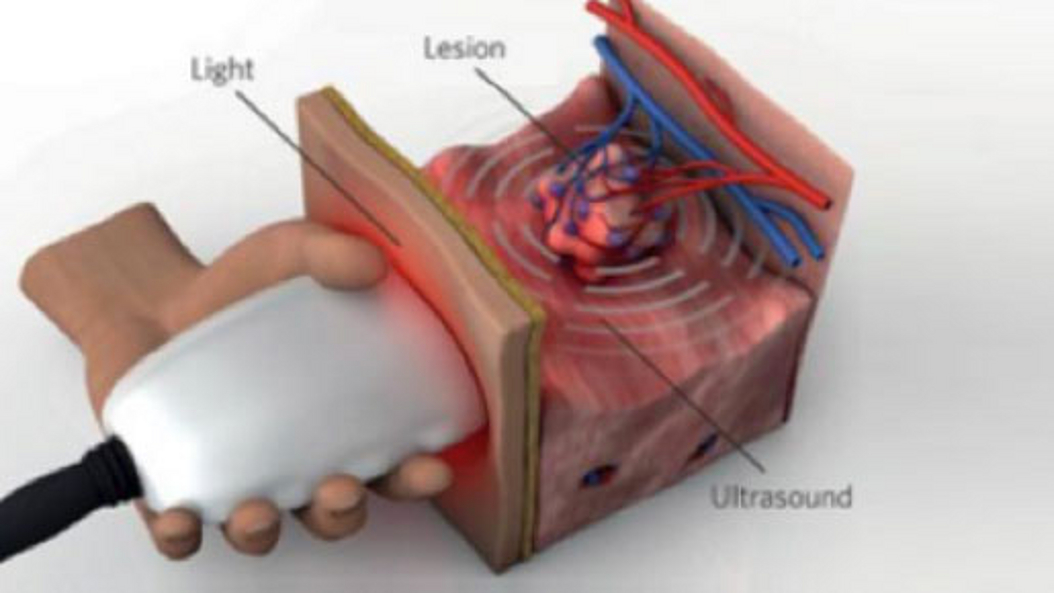
Optoacoustic imaging, or photoacoustic imaging, is insensitive to photon scattering within biological tissue and, unlike conventional optical imaging methods, and makes high-resolution optical visualization deep within tissue possible. Recent advances in laser technology, detection strategies and inversion techniques have led to significant improvements in the capabilities of optoacoustic systems. A key empowering feature - pioneered at TUM - is the development of video-rate multispectral imaging in two and three dimensions, which offers fast, spectral differentiation of distinct photo-absorbing modalities.
Experimental Magnetic Resonance Imaging
PI Dimitrios Karampinos
Research Group Body Magnetic Resonance
Publications
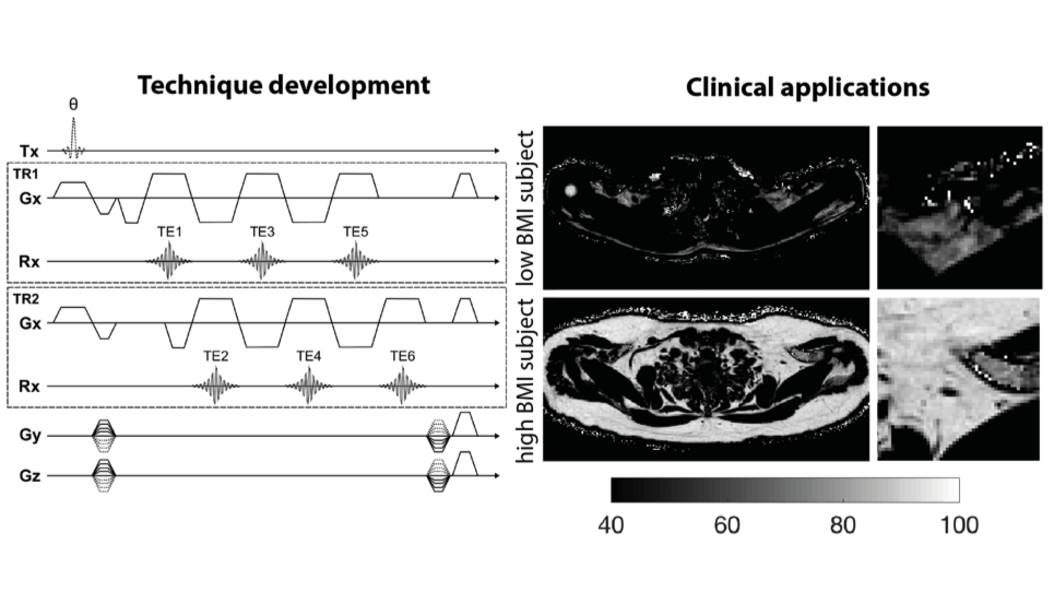
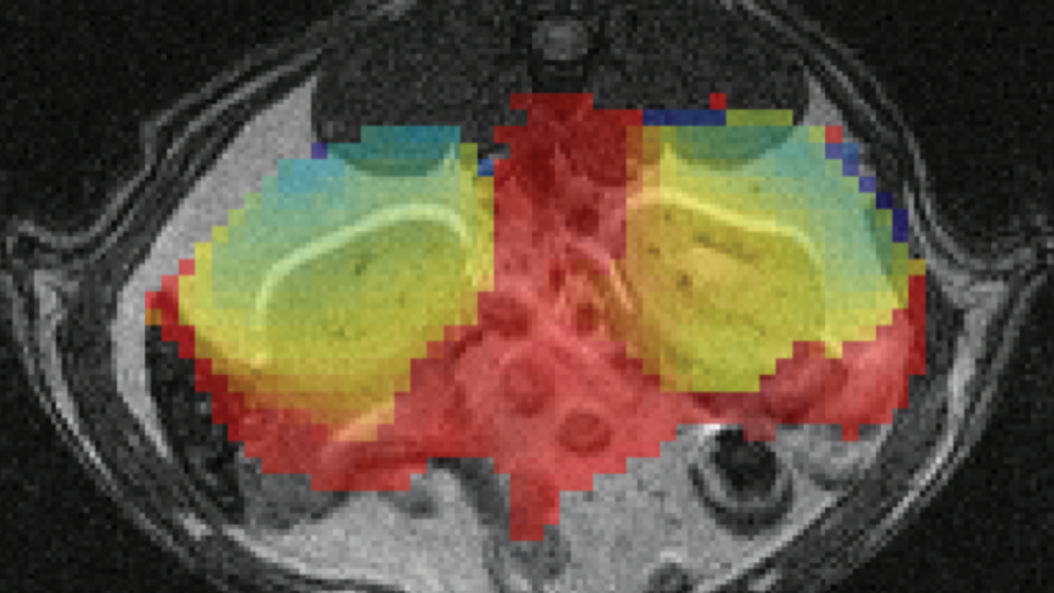
Prof. Karampinos' research focuses on the development of novel Magnetic Resonance Imaging (MRI) and Magnetic Resonance Spectroscopy (MRS) acquisition, reconstruction and signal modeling methods with an emphasis on the extraction of quantitative imaging biomarkers. He and his team are developing novel methods for proton density fat fraction mapping, lipid diffusion mapping, quantitative susceptibility mapping, T2 and T2* mapping, diffusion tensor imaging and body diffusion-weighted imaging. The developed methods are being translated into clinical studies for improving the diagnosis, the therapy monitoring, and the understanding of disease pathophysiology in the diseases of the musculoskeletal system (e.g. osteoporosis, neuropathies, neuromuscular diseases), in metabolic dysfunction (e.g. obesity, diabetes, cachexia) and in body oncology.
Magnetic Resonance Imaging Biomarkers
PI Franz Schilling
Research Group Biomedical Magnetic Resonance
Publications
Magnetic resonance imaging biomarkers enable a comprehensive characterization of tissue providing functional, physiological, metabolic, cellular and molecular information beyond anatomical structures. In recent years, hyperpolarization techniques allowed to increase the inherent low sensitivity of magnetic resonance by more than four orders of magnitude, opening new applications ranging from the tracking of chemical reactions in real-time to the direct monitoring of metabolic processes in vivo. Another functional MRI technique that has already shown promise in characterizing tissue in clinical studies is diffusion-weighted MRI (DW-MRI). It enables the quantitative assessment of the apparent diffusion coefficient (ADC) that depends on microstructural tissue properties, including cellularity and intercellular matrix composition. At TUM, sensitive hyperpolarized molecules and advanced diffusion MRI techniques are being developed aiming to provide novel imaging biomarkers to characterize tissue function.
X-Ray & Computed Tomography
Prof. Dr. Daniela Pfeiffer
Professorship of Diagnostic & Interventional Radiology
Publications
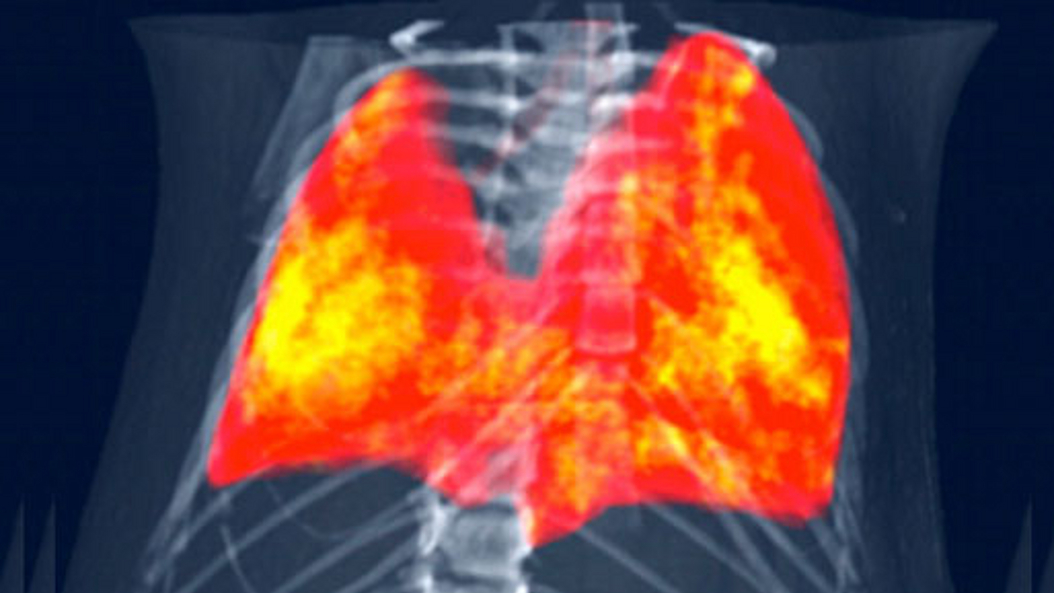
Daniela Pfeiffer is an expert in the field of lung imaging and musculoskeletal diagnostics. Her main medical research interest is improving detection, precise diagnosis and therapy follow-up of lung diseases such as COPD, lung cancer and infectious diseases (including COVID-19).
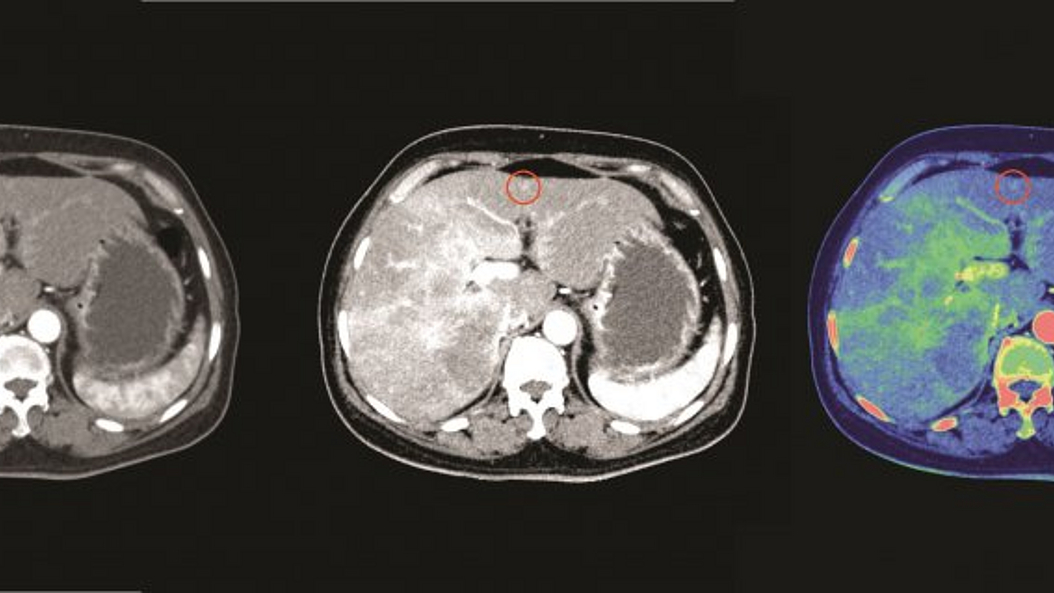
From a technical perspective, she is mainly interested in novel methods for medical X-Ray and Computed Tomography (CT) Imaging. Her main focus lies in the field of photon counting CT technology, spectral CT applications, dark-field and phase contrast imaging, and machine learning in clinical applications.
Diagnostic Radiology
Prof. Dr. Ernst Rummeny
Professorship of Radiology
Publications
Professor Rummeny was the director of the Institute for Radiology. His area of expertise is diagnostic radiology and nuclear medicine. The major focus of his research was in the field of oncology imaging, in particular the development of cross-sectional imaging processes such as MRI and CT tomography and linked hybrid systems, such as PET/CT and MR-PET, for the recognition and treatment assessment of malignant and benign disease. Together with colleagues from Biomedical Physics (Prof. F. Pfeffer) and Biological Imaging (Prof. V. Ntziachristos), he developed new devices for use in patient diagnosis. Furthermore, he was involved in the optimization of contrast medium.
Pharmaceutical Radiochemistry
Prof. Dr. Hans-Jürgen Wester
Chair of Pharmaceutical Radiochemistry
Publications
Prof. Wester’s research focuses on the development of targeted radiolabelled probes and therapeutics (radiopharmaceuticals) for molecular imaging and targeted radioligand therapy. He has developed several methodological procedures and radiopharmaceuticals for oncological, neurological and cardiovascular investigations and therapies that have found broad application in clinical investigations and therapies.