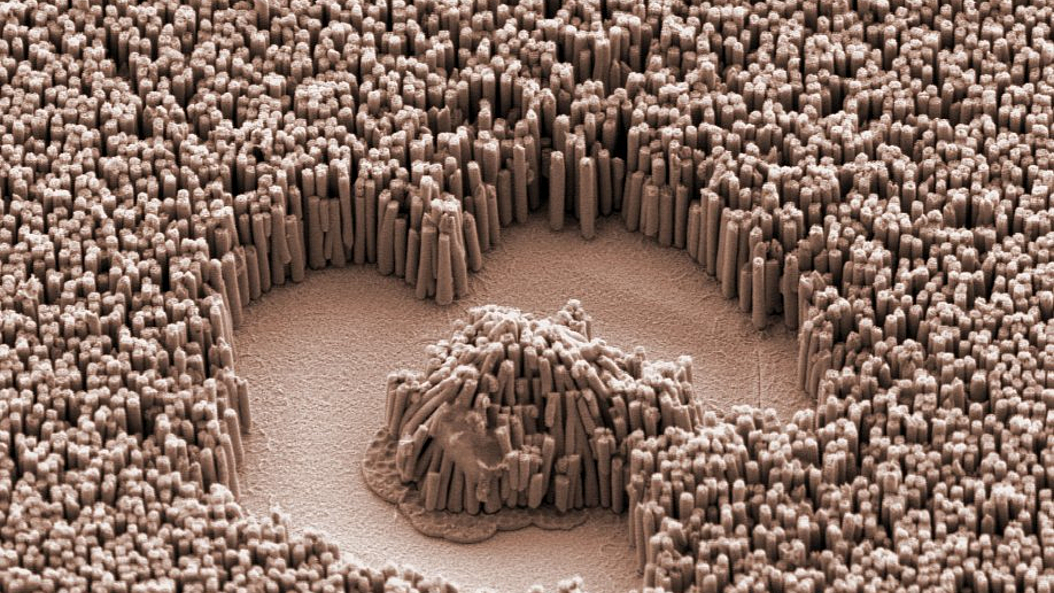
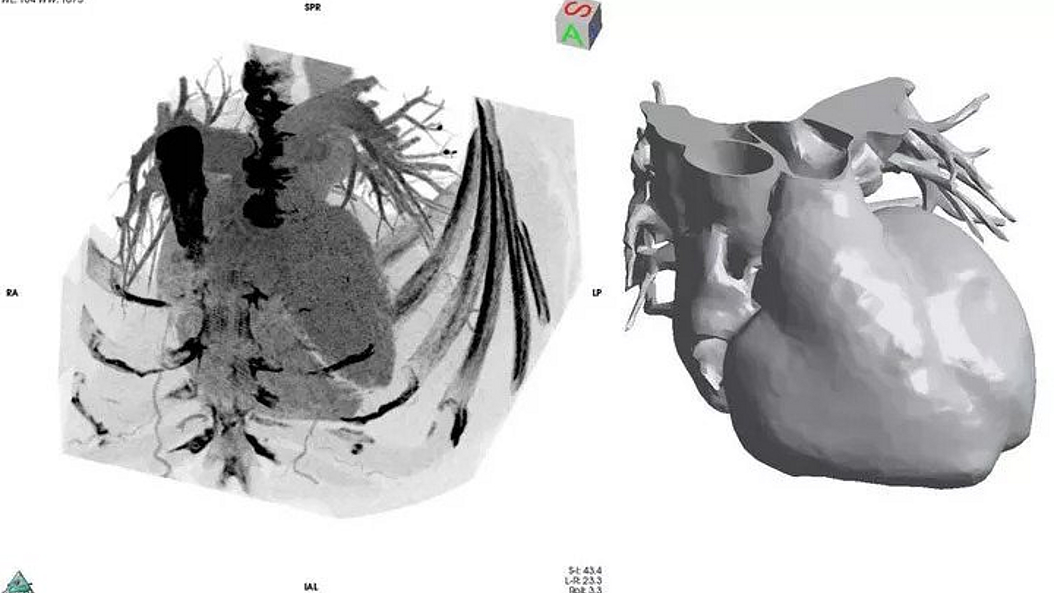
Biomaterials & Biomolecular Systems
In the research area Biomaterials & Biomolecular Systems, MIBE researchers investigate how the bio-physical principles underlying the formation and interaction of natural biological materials and biomolecular systems can be harnessed for diagnostic or therapeutic biomedical applications.
Molecular Devices & Machines
PI Prof. Hendrik Dietz
Research Group Biomolecular Nanotechnology
Publications
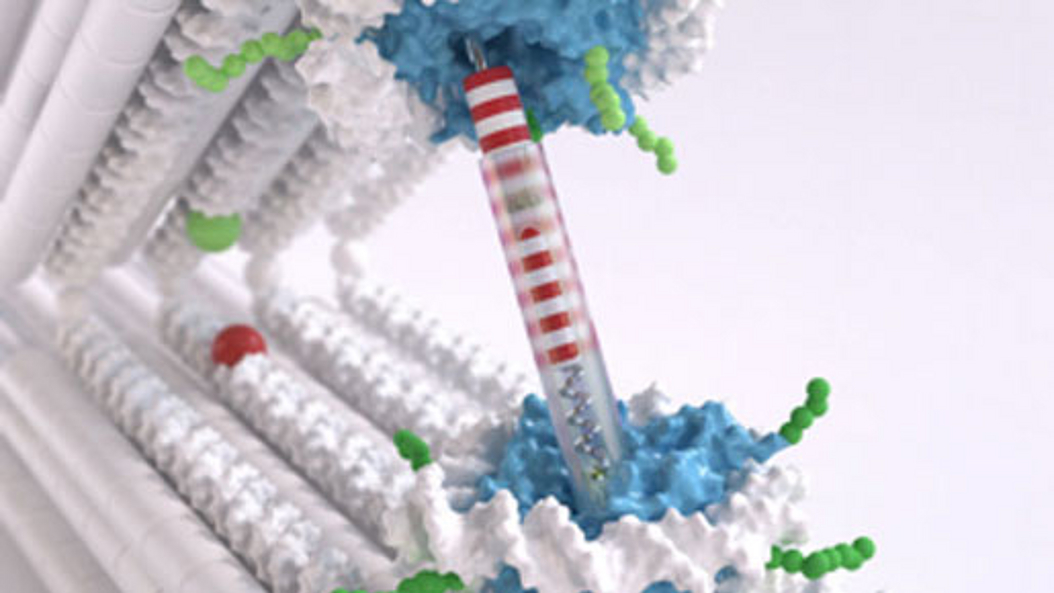
Inspired by the rich functionalities of natural macromolecular assemblies such as enzymes, molecular motors, and viruses, the team around Professor Dietz investigates how to build increasingly complex molecular structures. The goal is to build molecular devices and machines that can execute user-defined tasks. Molecular self-assembly with DNA is an attractive route toward achieving this goal. DNA origami in particular enables building nanodevices that can already be employed for making new discoveries in biomolecular physics and protein science.
Mammalian Cell Engineering
PI Gil Westmeyer
Research Group Molecular Imaging
Publications
It is of growing interest to decipher the patterns of cell-circuit signaling for understanding the (patho)physiology of living organisms. There is also a progression in biomedicine from applying small molecules and proteins to deploying genetically engineered cells as therapeutic agents in patients. Monitoring and controlling genetically defined cells in living organisms is thus of substantial importance. The research program of the team around Professor Westmeyer, therefore, focuses on bioengineering of next-generation molecular sensors and actuators for functional imaging and remote spatiotemporal control of cellular processes with whole‑organ(ism) coverage. They are, in particular, concentrated on gaining genetic control over key logistic processes in mammalian cells such as compartmentalization. In this way, they can install new metabolic pathways and generate self-assembling biomaterials with new, e.g., biomagnetic properties. These genetically controlled biophysical interfaces allow them to establish two-way communication with specific cells that they will bring to bear on future imaging-controlled tissue engineering and cell therapies.
Biopolymers & Biointerfaces
PI Oliver Lieleg
Research Group Biomechanics
Publications
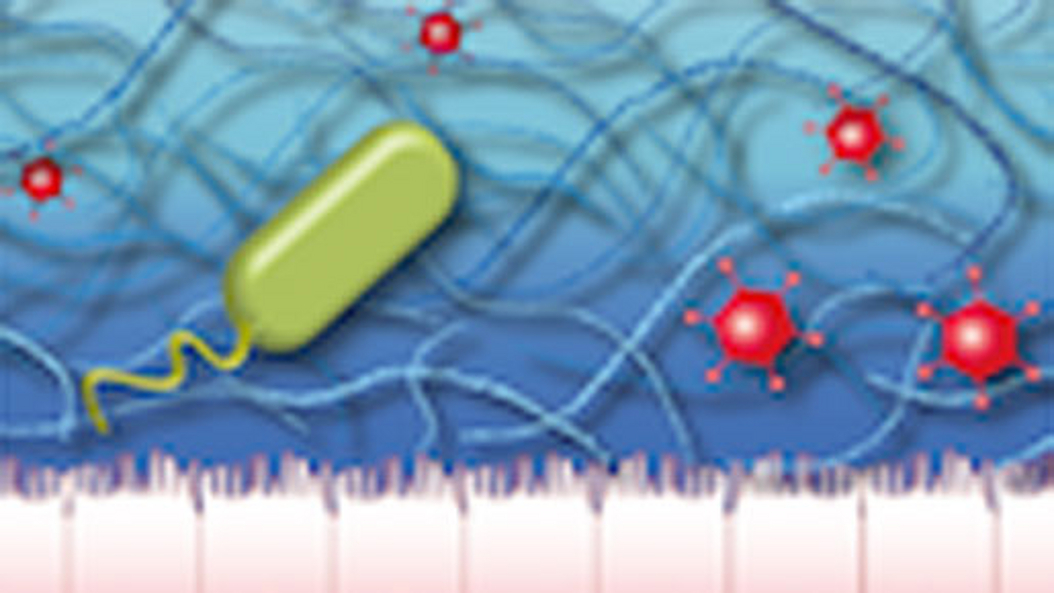
Biopolymers are located inside and outside of eukaryotic cells where they form hydrogels in aqueous environments. Examples include mucus, the extracellular matrix and bacterial biofilms, respectively. Such hydrogels have a dual function: First, they are responsible for the viscoelastic properties of cells and tissues and protect them from mechanical damage. Second, they regulate the passive transport of particles and molecules.
Professor Lieleg's research has the following goals:
- To discover new, to date unknown properties of biopolymers.
- To identify the microscopic principles that govern the material properties (e.g., mechanics, permeability, and lubricity) of biological hydrogels.
- To apply those principles to synthetic polymers, create biomimetic materials and find technical/medical applications for purified biopolymers
Creation of Synthetic Biological Systems
Prof Friedrich Simmel
Research Group Physics of Synthetic Biological Systems
Publications
The remarkable properties of biological systems are the result of complex interactions between multiple components, and thus emerge at the systems level. Biological systems are thus able to respond to their environment, compute, move, reconfigure and evolve. Professor Simmel aims at the construction of synthetic molecular and cellular systems, which generate and display similar behaviors. His team's work involves the engineering and study of synthetic gene circuits, the creation of artificial cellular systems and the development of cell-scale robots.
Neuroengineering Materials
PI Kristen Kozielski
Professorship of Neuroengineering
Spinal cord injuries, chronic pain or Parkinson’s disease – neural implants provide a potential solution to alleviate these neural injuries or diseases. Neural devices work by creating an electrical interface with the nervous system so that they can either measure or modulate neuronal activity. Surgery to install these implants, however, involves risk to the patient, such as infection or injury. Kristen Kozielski, Professor for Neuroengineering, and her team are working towards a potential alternative: Magnetoelectric nanoparticles that can serve as wireless neural implants. Instead of surgical implantation, the nanoparticles could be injected in the brain. First results show that injected nanoelectrodes are able to stimulate the brain in mice. Can the stimulation be more selective? Is neural stimulation able to mitigate negative effects caused by Parkinson’s disease? For how long would the effects last? And what happens to the nanoparticles over time? The research team is working to answer these questions by further improving, characterizing, and evaluating materials for neural nanoelectrodes.
Medical Materials & Implants
PI Petra Mela
Chair of Medical Materials and Implants
Publications
The Chair of Medical Materials and Implants (MMI) is offering several lectures, supplementary subjects and practical courses with a focus on biohybrid systems. Together with the Institute of Micro Technology and Medical Device Technology (MIMED), they are significantly involved in the Master's program Medical Engineering and Assistance Systems at the Department of Mechanical Engineering.
Nano- & Microrobotics
Prof. Dr. Berna Özkale Edelmann
Professorship for Nano- & Microrobotics
Publications
Berna Özkale Edelmanns Forschung konzentriert sich auf die Entwicklung nanorobotischer Werkzeuge zur Entschlüsselung von Signalprozessen zwischen Säugetierzellen und ihrer Mikroumgebung. Im Besonderen nutzt ihre Forschung Nanotechnologie, intelligente Biomaterialien, Mikrofluidik und Einzelzellmanipulationstechnologie, um multifunktionale Mikrogeräte für die regenerative Medizin zu entwickeln.
Computer Aided Plastic Surgery
Prof. Dr. med Laszlo Kovacs
Research Group Computer Aided Plastic Surgery
Publications
Prof. Dr. Laszlo Kovacs from the Department of Plastic Surgery and Hand Surgery (Klinikum rechts der Isar der Technischen Universität München) initiated the research group Computer Aided Plastic Surgery (CAPS).
The leading idea of this group is the fusion of various disciplines e.g. bioengineering, mathematics, physics, biomechanics, computer science and other associated disciplines in order to create new knowledge and solutions with benefit for interdisciplinary problems.
The scientific focus of CAPS evaluates innovative technologies for the 3-D assessment, digitalization and visualisation of the human body surface and soft tissues to implement computer aided surgery (CAS) into the field of aesthetic, plastic and reconstructive surgery.